More than 60% of regulatory observations in pharmaceutical manufacturing relate to documentation control, investigation quality, and procedural compliance gaps. GMP Interview preparation in regulated environments determines whether a candidate can operate safely under inspection pressure. Industry hiring cycles for quality and production roles often span 6–12 weeks, and a single weak decision may later surface during an audit or deviation review. Therefore, preparation must go beyond memorizing definitions. Instead, professionals must demonstrate structured thinking, documentation discipline, and regulatory awareness. Compliance pressure continues to increase across manufacturing sites that operate under Good Manufacturing Practices (GMP).
Regulatory agencies focus on human error, incomplete records, and weak investigations; therefore, interview panels simulate real operational scenarios to assess how candidates respond to documentation gaps and escalating deviations, helping them align answers with inspection logic rather than generic advice.
Table of Contents
What is a GMP Interview in a Pharmaceutical Regulatory Context
A regulatory-focused interview evaluates whether a professional can protect product quality, maintain traceable documentation, and make defensible decisions under inspection pressure. Instead of testing definitions, panels assess how candidates explain deviation handling, documentation control, escalation logic, and role accountability within quality systems. Because pharmaceutical manufacturing operates under strict oversight, interviewers look for structured reasoning and inspection-ready thinking that aligns daily actions with controlled procedures and documented evidence.
How Interview Questions Reflect Real Inspection Expectations
Interview questions reflect inspection logic because companies hire to control compliance risk. When regulators review a site, they follow documented evidence step by step; therefore, hiring managers assess whether candidates think in the same structured way. Facilities with complex processes and strict oversight face the highest hiring pressure. As a result, panels focus on how candidates handle deviations, protect data integrity, and escalate issues. They expect clear explanations of what happened, which record supports the action, who approved the decision, and how recurrence will be prevented. Structured answers signal inspection readiness, while vague responses raise concern.
High-Impact Question Areas for Inspection Readiness
The following visual map summarizes the key competency domains that interview panels evaluate to predict inspection performance.
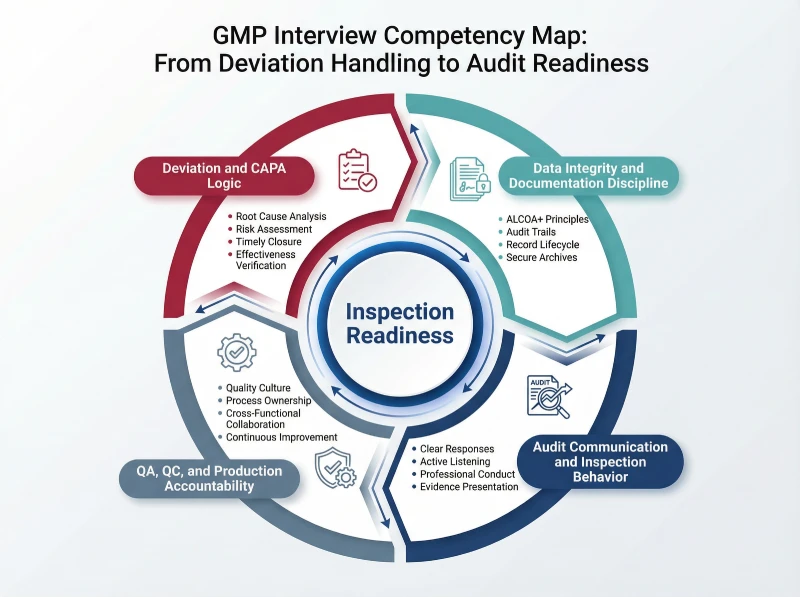
Regulated manufacturing requires defensible decisions, so interview panels focus on documentation discipline, deviation logic, and accountability to assess inspection readiness under regulatory pressure.
The following domains represent the most critical evaluation areas aligned with inspection expectations:
- Deviation and CAPA Interview Questions in GMP Roles
- Data Integrity and Documentation Interview Questions GMP
- GMP Audit and Inspection Scenario Questions
- QA, QC, and Production GMP Interview Questions Explained
Deviation and CAPA Interview Questions in GMP Roles
Panels frequently test how candidates handle deviations because investigations directly affect compliance outcomes. Therefore, professionals must explain containment first, then impact assessment, and finally corrective action logic. Strong answers show ownership, structured documentation, and clear escalation. Moreover, candidates should describe how they verify CAPA effectiveness, since recurrence control matters more than initial reaction.
Data Integrity and Documentation Interview Questions GMP
Interviewers examine documentation discipline because inspectors rely on records, not intentions. Consequently, candidates must demonstrate contemporaneous recording, traceable corrections, and controlled data handling. In addition, they should explain how they protect audit trails and escalate missing information. Clear, evidence-based explanations signal inspection readiness, while vague responses create risk.
GMP Audit and Inspection Scenario Questions
Audit scenarios test composure and factual communication. Therefore, candidates should respond with documented facts, defined roles, and clear next steps. They must avoid speculation and instead describe how they verify information through controlled procedures. Because inspectors follow the evidence chain, structured responses demonstrate operational maturity.
QA, QC, and Production GMP Interview Questions Explained
Each function carries distinct accountability. QA governs decisions and approves investigations, QC controls analytical data, and Production executes procedures and records actions. Therefore, candidates must clarify boundaries and escalation pathways. When professionals articulate role-specific responsibility clearly, they strengthen credibility and reduce perceived compliance risk.
Common GMP Interview Questions and Structured Answer Framework
Interview panels repeatedly use high-frequency questions to test regulatory thinking, documentation discipline, and deviation logic. Therefore, candidates must prepare structured responses that align with inspection expectations.
The table below highlights eight high-search and high-impact questions frequently asked in pharmaceutical interviews.
Common Question | What It Tests | Structured Approach |
How do you handle a deviation in manufacturing? | Ownership and escalation logic | Contain, assess impact, document, investigate, implement CAPA |
What is data integrity and why does it matter in manufacturing? | Documentation discipline | Traceability, contemporaneous entry, audit trail awareness |
How do you manage CAPA effectively? | Prevention mindset | Root cause analysis, corrective action, effectiveness verification |
What would you do if a batch failed specifications? | Risk management | Quarantine, investigation, impact assessment, decision authority |
How do you prepare for a regulatory audit? | Inspection readiness | Review records, verify procedures, confirm role accountability |
What is the difference between QA and QC responsibilities? | Role accountability | Governance authority versus analytical control |
These questions reflect real operational pressure points. Moreover, candidates who answer in structured sequences demonstrate inspection-aligned thinking. In contrast, vague or overly theoretical responses reduce credibility. Therefore, preparation should focus on decision logic, documentation clarity, and escalation pathways
Building an Inspection-Oriented GMP Interview Preparation Plan
The following framework illustrates how structured responses align with inspection expectations in regulated manufacturing environments.
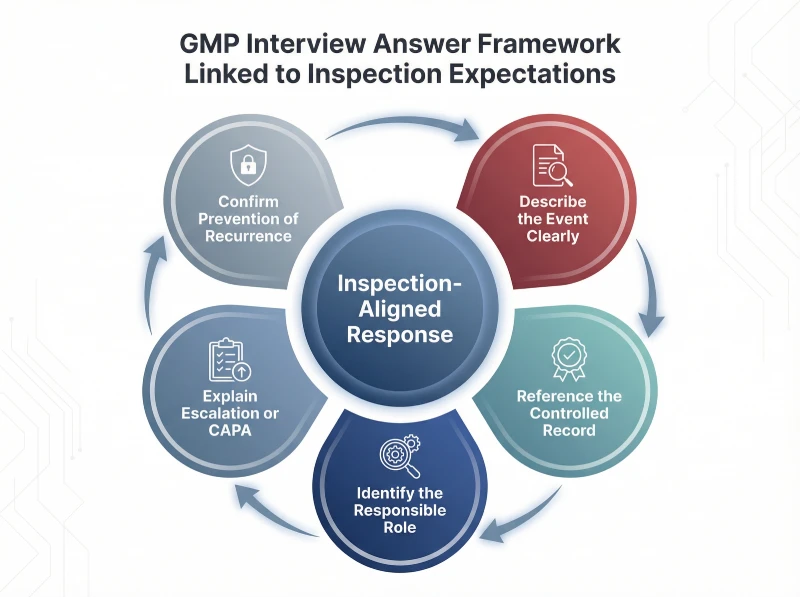
Preparation reduces variability and strengthens confidence under regulatory questioning. Therefore, professionals should practice structured examples that reflect real manufacturing scenarios rather than abstract definitions. Consistent rehearsal improves clarity and minimizes hesitation.
The table below outlines a focused preparation structure aligned with inspection expectations.
Preparation Step | Focus Area | Expected Benefit |
Map real examples | Deviations and CAPA | Clear narratives |
Review documentation principles | Data integrity | Fewer probing gaps |
Simulate audit questions | Inspection demeanor | Confident responses |
Clarify role boundaries | QA vs QC vs Production | Accurate escalation |
GMP Competency Framework Aligned with Regulatory Inspection Expectations
A focused weekly routine drafting deviation cases, refining data integrity explanations, rehearsing audit scenarios, and clarifying escalation roles strengthens decision clarity and operational discipline.
Final Word
In recent FDA inspection cycles, more than 30% of Form 483 observations relate directly to documentation practices and investigation deficiencies. This statistic highlights a clear operational reality: companies prioritize professionals who can defend their decisions with documented evidence and structured reasoning. Therefore, preparation for a GMP Interview must align with real inspection pressure rather than memorized answers.
Candidates who explain deviation handling, CAPA logic, and escalation pathways in clear sequences demonstrate operational maturity. Moreover, those who rehearse inspection-style scenarios respond with greater confidence and clarity under technical questioning.
If you would like structured guidance, scenario-based practice, and focused preparation aligned with regulated manufacturing expectations, you can access interview readiness support through a PharmUni Pro subscription. To review available options and compare plans, visit the Pricing page.
FAQs
Hiring panels usually focus on deviation handling, documentation control, data integrity principles, CAPA management, and audit readiness. These topics reflect daily responsibilities in regulated manufacturing environments.
Because inspection findings often stem from weak investigations or incomplete records. Strong answers show structured thinking, traceable documentation, and clear escalation pathways.
Use a structured sequence: explain the event, reference the controlled record, define approval responsibility, and describe how recurrence is prevented. This approach aligns with real inspection expectations.

Mahtab Shardi
Mahtab is a pharmaceutical professional with a Master’s degree in Physical Chemistry and over five years of experience in laboratory and QC roles. Mahtab contributes reliable, well-structured pharmaceutical content to Pharmuni, helping turn complex scientific topics into clear, practical insights for industry professionals and students.

Pharma Jobs in Pittsburgh in 2026: GMP Careers, Hiring Trends and Industry Demand
This article explores pharmaceutical jobs in Pittsburgh, focusing on GMP compliance careers, QA/QC roles, validation engineer opportunities, and FDA regulated industry jobs. It highlights hiring trends, skill requirements, and growth pathways across pharmaceutical manufacturing and quality systems.

pharma jobs in USA in 2026 GMP Careers and Hiring Trends Guide
This article explores pharma jobs in USA, focusing on hiring trends, GMP careers, salary growth, and key roles in clinical research and regulatory affairs. It helps candidates understand how the U.S. pharmaceutical job market is evolving today.

Process Validation statistics in 2026: GMP data analysis guide
This article explains how pharmaceutical process validation metrics and statistical tools improve GMP compliance through Cp Cpk analysis, control charts, and PPQ batch performance statistics. It highlights how data-driven validation increases process stability, reduces variability, and supports inspection readiness.