More than 40% of recent FDA warning letters referencing manufacturing systems have included data integrity observations, highlighting how frequently regulators identify weaknesses in electronic records and master data governance. Data in GMP remains one of the most scrutinized areas during inspections, especially as sites rely increasingly on computerized systems.
As digital infrastructure expands, regulators examine audit trails, access control models, and change management workflows in greater depth. For regulated manufacturers, the challenge is no longer documentation volume; instead, it is documentation reliability under inspection pressure. This article examines how structured governance strengthens data integrity and protects compliance under Good Manufacturing Practices (GMP).
Table of Contents
What Is Data in GMP in a Pharmaceutical Manufacturing Context
It defines how regulated manufacturers create, control, review, and protect operational and master data under inspection expectations.
Manufacturing sites generate large volumes of batch records, specifications, equipment settings, and quality decisions. Regulators expect this information to remain complete, attributable, and accurate under ALCOA principles. Therefore, teams must ensure that every recorded value and system change stays traceable and defensible during inspection.
Operational data supports daily production, while master data defines critical parameters such as specifications and system configurations. Because master data management in pharma directly affects downstream processes, even small inaccuracies can spread quickly. Consequently, organizations must treat master data lifecycle control as a core quality responsibility, not just an IT function.
Why Data in GMP Governance Drives Inspection Outcomes
Inspection outcomes depend on how clearly a site demonstrates control, traceability, and accountability across its data systems. Regulators review audit trails, user access structures, and change management workflows to assess data integrity in pharmaceutical manufacturing. Therefore, if electronic records lack transparency, inspectors expand scope quickly.
Transparent audit trails, controlled master data, and structured change governance form the foundation of defensible compliance.
Strong governance aligns electronic batch records with validated systems and documentation control. However, when user roles overlap or change control lacks justification, regulators question overall quality oversight.
Data in GMP Governance Maturity Levels for Inspection Readiness
Inspection readiness strengthens as governance progresses from basic master data control to structured lifecycle and change oversight. Organizations that define ownership, enforce audit transparency, and integrate change management into daily operations reduce regulatory risk significantly. As maturity increases, data integrity shifts from reactive correction to proactive control.
The model below illustrates how governance maturity progresses from basic master data control to full lifecycle and change oversight.
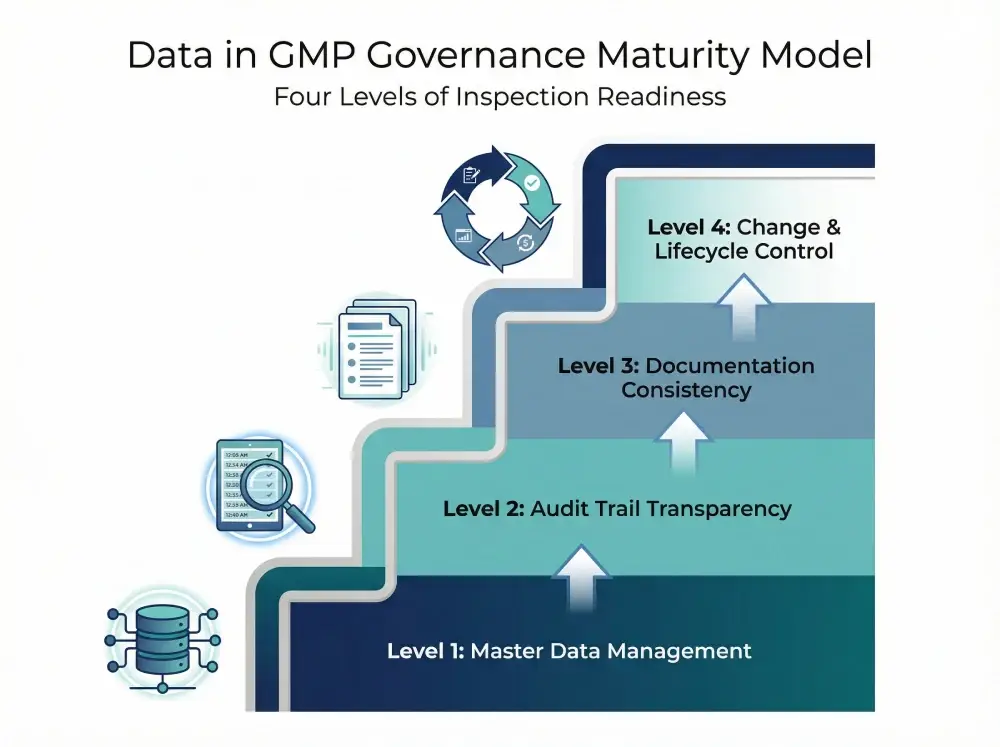
The four maturity levels below outline how governance evolves across operational and master data environments:
- Level 1: Master Data Management in Pharmaceutical Manufacturing
- Level 2: Audit Trail and Electronic Record Transparency
- Level 3: Documentation Control and System Consistency
- Level 4: Change Governance and Data Lifecycle Control
Level 1: Master Data Management in Pharmaceutical Manufacturing
At the foundation, teams define ownership for product specifications, material codes, and system parameters. They control updates through formal approval workflows. In inspections, auditors frequently compare master data configuration against approved specifications; inconsistencies immediately raise concern.
Level 2: Audit Trail and Electronic Record Transparency
Next, sites ensure that audit trails remain enabled, reviewed, and protected from alteration. Transparent logs document who changed what and why. During inspections, regulators often request raw audit trail extracts rather than summary reports; therefore, review discipline becomes critical.
Level 3: Documentation Control and System Consistency
At this stage, documentation control integrates with validated systems. SOP versions align with electronic workflows, and training matrices match system permissions. When documentation and system behavior diverge, compliance credibility weakens.
Level 4: Change Governance and Data Lifecycle Control
At the highest level, organizations govern data across its lifecycle—from creation through archival. Structured change control links impact assessment, validation updates, and documentation revision. Because data modifications affect product quality directly, disciplined lifecycle governance reduces regulatory exposure.
Recurrent Data Integrity Findings in Regulated Manufacturing Sites
Most inspection findings stem from weak control linkage rather than missing procedures.
Common deficiencies relate to disabled audit trails, shared user accounts, uncontrolled master data changes, and inadequate documentation review.
The table below summarizes typical findings and their root causes.
Inspection Finding | Root Cause | Regulatory Risk | Expected Control |
Disabled or unreviewed audit trails | Lack of periodic review | Loss of traceability | Routine audit trail review procedure |
Shared login credentials | Weak access governance | Accountability gaps | Unique user IDs with role control |
Uncontrolled master data updates | Informal change process | Parameter inconsistency | Approved change workflow with validation impact |
Documentation mismatch | SOP not aligned with system | Process deviation risk | Integrated documentation control |
Incomplete electronic batch records | Manual override without justification | Data reliability concern | Structured review and approval checkpoints |
These patterns reveal a central theme: regulators expect integrated pharmaceutical data governance, not isolated IT controls.
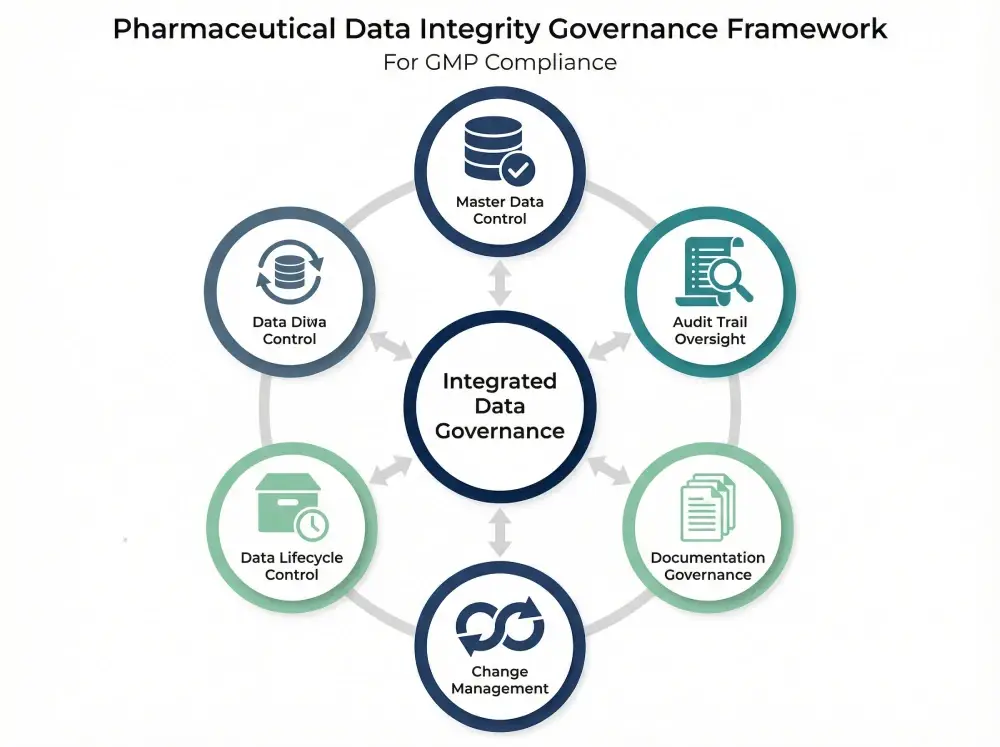
Building a Pharmaceutical Data Integrity Governance Framework
An effective governance framework connects master data control, audit transparency, and documentation discipline under unified quality oversight. Organizations must assign clear ownership of master data, enforce structured approval workflows, and conduct periodic review. In addition, teams should actively review audit trails and document anomaly assessments.
Change governance must link every system modification to impact evaluation and validation updates. Finally, lifecycle control ensures reliable data from creation through archival. When these elements operate together, pharmaceutical data governance becomes inspection-ready rather than reactive.
The framework below illustrates how master data control, audit transparency, and change governance integrate into a unified compliance structure.
Final Word
In fiscal year 2023 alone, the FDA conducted more than 1,600 drug quality surveillance inspections worldwide, reflecting the scale and intensity of regulatory oversight in pharmaceutical manufacturing. As inspection frequency remains high, data integrity failures continue to surface under routine review, particularly in audit trail transparency and master data control.
For Data in GMP, structured governance protects regulatory timelines because it ensures transparent traceability, accountable change control, and validated system behavior. In real inspections, auditors often request raw audit trail extracts and master data configuration records within minutes of initiating system review. Therefore, sites that lack disciplined governance quickly lose control of the inspection narrative.
When master data management, documentation control, and audit transparency operate as one integrated system, compliance shifts from reactive correction to sustained operational control.
FAQs
Unreviewed audit trails, shared user accounts, undocumented data changes, and inconsistencies between electronic batch records and approved specifications commonly trigger findings. Inspectors focus on traceability and accountability in computerized systems.
Audit trails provide evidence of who changed data, when, and why. Without routine review and controlled access, regulators cannot verify compliance with ALCOA principles or ensure reliable documentation control.
Assign clear ownership of master data, enforce unique user access, conduct periodic audit trail reviews, and link every system change to documented impact assessment and validation updates. Consistent oversight reduces regulatory risk.

Mahtab Shardi
Mahtab is a pharmaceutical professional with a Master’s degree in Physical Chemistry and over five years of experience in laboratory and QC roles. Mahtab contributes reliable, well-structured pharmaceutical content to Pharmuni, helping turn complex scientific topics into clear, practical insights for industry professionals and students.

Pharma Jobs Los Angeles in 2026: GMP Compliance and Salary Trends
Los Angeles continues to expand across pharmaceutical companies in Los Angeles and biologics manufacturing Southern California. From GMP jobs in Los Angeles to pharmacist jobs Los Angeles CA, hiring demand reflects strong regulatory oversight and advanced clinical research growth in LA – California.

Master ICH Q7 Guideline in 2026: Essential API GMP Rules You Must Know
ICH Q7 is the GMP guide for active pharmaceutical ingredients (APIs). It sets detailed standards for quality, documentation, validation, and supplier control. WHO reports that poor supplier management causes up to 30% of API quality issues. Following ICH Q7 ensures consistent product safety and regulatory compliance worldwide.

Pharma Jobs in Atlanta in 2026: Compliance and Salary Landscape
Atlanta’s regulated life sciences market reflects growing demand across pharmacist jobs in Atlanta, biotech jobs in Atlanta, and clinical research jobs in Atlanta. Within FDA regulated companies in Georgia, employers prioritize compliance expertise, validation accountability, and structured quality systems experience.