Pharmaceutical Warehouse compliance often fails where temperature data, segregation status, and inventory records do not align—especially during routine inspections or deviation-driven follow-ups. In 2019, a cross-industry review of FDA Form 483 observations identified more than 155 environmental monitoring deficiencies, most linked to temperature control; therefore, storage oversight remains a recurring inspection trigger.
For QA and warehouse leaders, the issue rarely stems from missing SOPs. Instead, gaps appear when mapped zones do not guide probe placement, when quarantine status conflicts with floor reality, or when teams log excursions without clear impact assessment. Consequently, inspection defensibility depends on how well operations connect daily control to documented evidence under Good Manufacturing Practices (GMP).
Table of Contents
What Is a Pharmaceutical Warehouse in a GMP Context
GMP warehouse requirements define a regulated control framework that safeguards product quality during storage and distribution. Regulators treat warehousing as an extension of manufacturing; therefore, facilities must actively control environmental conditions, enforce strict segregation between approved and non-approved materials, and maintain accurate batch traceability. In addition, teams must qualify storage areas, monitor temperature continuously, and ensure that system status always matches physical stock. Finally, operations must manage dispatch and transport interfaces under GDP expectations so environmental control and documentation remain consistent across the supply chain.
Why Pharmaceutical Warehouse Controls Drive Inspection Outcomes
Warehouse controls determine inspection outcomes because inspectors evaluate storage discipline as a direct indicator of overall GMP and GDP compliance. Storage conditions for pharmaceuticals must remain within validated limits through mapped areas, continuous monitoring, and documented excursion assessment. At the same time, segregation between approved, quarantined, rejected, and returned materials must prevent mix-ups and align with system status. In addition, accurate records and batch traceability must support rapid verification during walkthroughs. When environmental data, segregation logic, and documentation align, the operation demonstrates drug storage compliance; however, when they diverge, regulators quickly expand the scope of review.
Pharmaceutical Warehouse Control Maturity Levels for Inspection Readiness
Inspection readiness does not happen at once; instead, it develops as storage controls evolve from basic environmental monitoring to fully integrated governance across segregation, traceability, and distribution. Sites that understand this maturity progression can identify weak control layers early and strengthen them before inspections expose gaps. In practice, regulators evaluate not only whether controls exist, but also how consistently teams execute and review them over time.
Before reviewing each level in detail, the infographic below summarizes how these control layers build on one another and strengthen inspection readiness step by step.
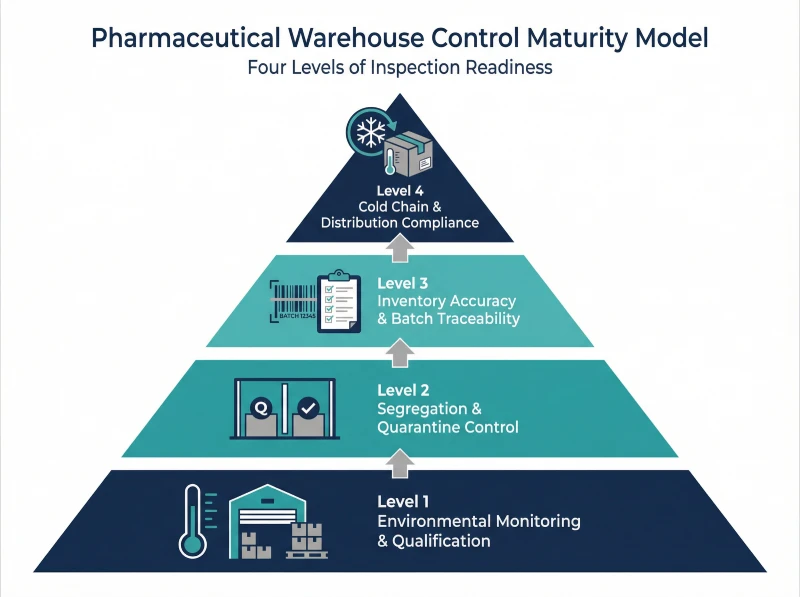
In the following section, we outline the four maturity levels that define how storage control systems progress toward inspection defensibility.
- Level 1: Environmental Monitoring and Warehouse Qualification
- Level 2: Segregation and Quarantine Area Control
- Level 3: Inventory Accuracy and Batch Traceability
- Level 4: Cold Chain and Distribution Interface Compliance
Level 1: Environmental Monitoring and Warehouse Qualification
At the foundation, environmental control defines compliance stability. Teams conduct risk-based temperature mapping, monitor conditions continuously, and assess excursions to maintain defensible storage oversight.
Level 2: Segregation and Quarantine Area Control
Building on environmental stability, segregation protects product status integrity. Clear separation of approved and non-approved materials, aligned with system records, prevents mix-ups and unintended release.
Level 3: Inventory Accuracy and Batch Traceability
Once status control is secure, traceability strengthens operational governance. Regular cycle counts and prompt discrepancy review ensure fast batch tracking during recalls or inspections.
Level 4: Cold Chain and Distribution Interface Compliance
Finally, control must extend beyond the warehouse walls. Teams manage transport conditions and review shipment data to preserve product integrity under GDP expectations.
Regulatory Gaps Frequently Observed in Pharmaceutical Warehouses
Inspectors most often cite gaps in temperature control, segregation discipline, and inventory integrity. Typically, the issue is not missing procedures but weak alignment between operations and documentation. When mapping does not guide monitoring, when quarantine status does not match system records, or when discrepancies remain unresolved, regulators question overall control. Because inspectors compare data with floor reality, small inconsistencies can quickly escalate.
The table below summarizes the most frequent inspection gaps and the expected control standard.
Inspection Gap | Operational Breakdown | Regulatory Risk | Expected Control Standard |
Mapping not linked to monitoring | Sensors lack risk-based placement | Undetected temperature extremes | Risk-driven probe strategy and trend review |
Excursions without assessment | Logged but not evaluated | Product impact uncertainty | Documented impact analysis and disposition |
Weak quarantine segregation | Mixed-status materials | Unintended release risk | Strict physical and system status alignment |
Documentation inconsistencies | Incomplete or delayed records | Data credibility concerns | Contemporaneous, reviewed documentation |
Stock discrepancies | Weak reconciliation discipline | Traceability delays | Investigated discrepancies and reconciled inventory |
Designing an Inspection-Ready Pharmaceutical Warehouse Control Structure
Inspection-ready storage requires integration. Sites must align monitoring, segregation, and inventory governance under clear ownership and review. When these controls operate together, they create defensible inspection evidence.
The framework below shows how these layers connect within a unified governance model.
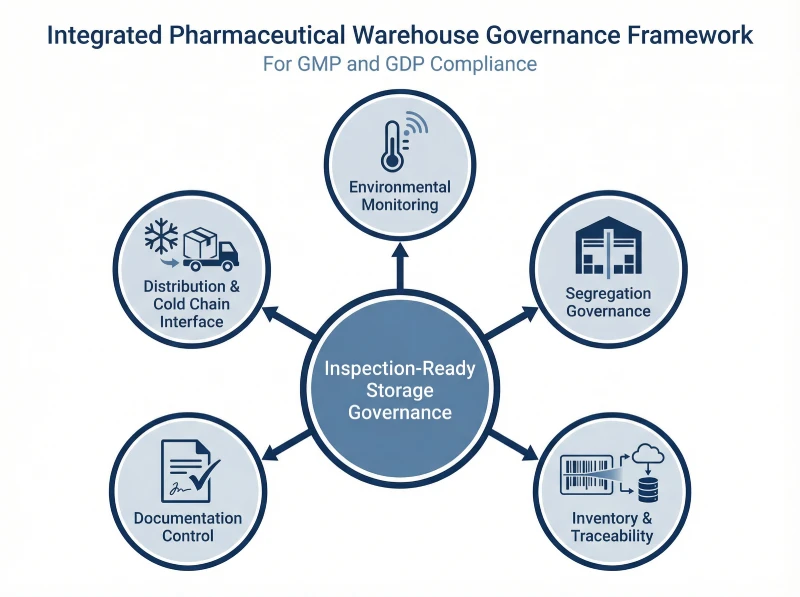
The table below summarizes the key control layers and required inspection evidence.
Control Layer | Responsible Function | Core Control Activities | Inspection Evidence |
Environmental Monitoring | Engineering / QA | Mapping, calibration, alarm review | Trend data and qualification reports |
Segregation Governance | Warehouse / QA | Status control, quarantine management | Floor-to-system reconciliation |
Inventory Integrity | Supply Chain / QA | Cycle counts, discrepancy review | Batch traceability documentation |
Documentation Control | QA | SOP lifecycle and record review | Controlled, contemporaneous records |
Distribution Interface | Logistics / QA | Shipment monitoring and excursion workflow | Documented release decisions |
When these layers align, the warehouse supports product quality and inspection credibility through structured, reviewable control.
Final Word
In global inspection cycles, regulators still report high rates of storage and distribution weaknesses, especially around temperature excursions and quarantine discipline. For example, a review of more than 99 GMP inspection reports across 19 countries identified 1,458 deficiencies, with nearly 37% classified as major issues many of which related to storage and environmental controls.
For a Pharmaceutical Warehouse, structured governance protects product quality because it prevents evidence gaps between environmental data, segregation status, and inventory records. Moreover, it stabilizes regulatory timelines by ensuring teams assess deviations promptly and document release decisions clearly. When monitoring, traceability, and documentation align consistently, the warehouse strengthens inspection credibility and reduces the risk that minor storage issues expand into broader compliance findings.
FAQs
In regulated drug storage, repeated or unassessed temperature excursions trigger findings. Inspectors expect defined alert and action limits, documented impact assessments, and clear disposition decisions for every deviation.
Because inspectors use segregation control as proof of release discipline. If system status does not match physical stock, or if returned goods lack documented evaluation, regulators question overall governance and GDP alignment.
Map risk-based temperature zones, reconcile physical and electronic status daily, investigate stock discrepancies promptly, and document excursion impact decisions clearly. Consistent linkage between monitoring, segregation, and traceability builds defensible compliance.

Mahtab Shardi
Mahtab is a pharmaceutical professional with a Master’s degree in Physical Chemistry and over five years of experience in laboratory and QC roles. Mahtab contributes reliable, well-structured pharmaceutical content to Pharmuni, helping turn complex scientific topics into clear, practical insights for industry professionals and students.

Audit Trail Review: Complete Guide to GMP Requirements, SOP Steps, and Inspection Readiness in 2026
Audit Trail Review is a critical process in pharmaceutical manufacturing that involves examining electronic records to ensure all entries, changes, and deletions are accurately documented and traceable. This review maintains data integrity, prevents errors, and detects unauthorized activities.

Pharma Jobs in Switzerland in 2026: Salary, Licensing, and Hiring Demand
Switzerland offers structured career paths across hospital, industry, and regulatory roles. This guide explains pharmacy salary in Switzerland, Switzerland pharmacist license requirements, and hiring demand within the Basel life sciences cluster and multinational pharmaceutical employers.

Release for Distribution in Pharma Manufacturing: a 2026 Guide on GMP Controls, Roles, and Required Documents
Release for Distribution requires Quality Unit authorization confirming all specifications met per WHO GDP Chapter 8, alongside logistics verification of temperature control, documentation, and shipping readiness before patient delivery. This dual authorization ensures regulatory compliance and product integrity throughout the supply chain. Understanding these processes prevents delays and protects patient safety by maintaining GMP standards and traceability requirements.