In recent global inspection cycles, a substantial share of Pharmacovigilance findings often estimated at over 30% in multi-region audits has been linked to inconsistent safety practices across countries. These observations show why harmonization in pharmacovigilance has shifted from a strategic ambition to a recurring inspection concern. Regulators increasingly interpret fragmented global safety operations as signs of weak control, especially when the same product generates divergent reporting, assessment, or signal management outcomes across regions.
This article examines how inspectors evaluate alignment across global safety systems, why lack of harmonization repeatedly triggers inspection findings, and how organizations can meet regulatory expectations without sacrificing justified local adaptation.
Table of Contents
What harmonization means in a regulatory safety context
Harmonization means demonstrating consistent control of safety activities across regions while allowing justified local variation, a core expectation increasingly described by regulators as harmonization in pharmacovigilance.
From an inspection perspective, regulators do not expect identical processes everywhere. Instead, they look for clear alignment of principles, decision logic, and governance across global pharmacovigilance operations.
In practice, harmonization refers to the ability of an organization to apply the same safety standards, assessment criteria, and escalation pathways across markets, even when operational details differ. Inspectors therefore focus on whether global oversight mechanisms ensure that safety decisions remain comparable, traceable, and defensible regardless of where data originates.
Crucially, harmonization is assessed through outcomes, not frameworks. When inspectors observe consistent case assessment quality, aligned signal decisions, and comparable reporting behavior across regions, they interpret this as evidence of effective regulatory convergence rather than superficial standardization.
Why inconsistent safety practices trigger inspection findings
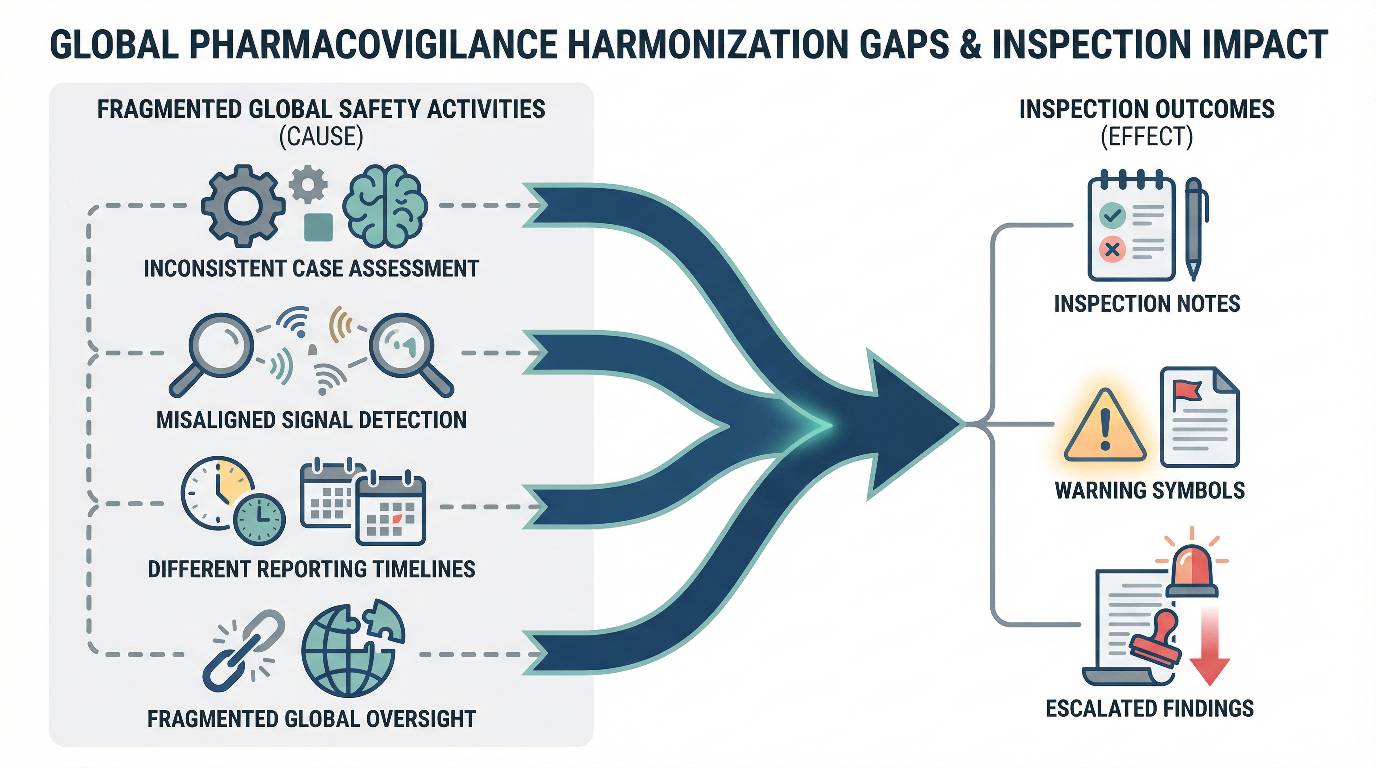
Inconsistent safety practices create inspection risk because they undermine regulatory confidence in system control, particularly when inspectors assess whether harmonization in pharmacovigilance is effectively embedded across regions.
When the same safety issue produces different conclusions, timelines, or actions across markets, inspectors question whether decisions are driven by global governance or by local interpretation. During inspections, authorities often compare how similar adverse events are handled in different regions.
Divergent causality assessments, reporting thresholds, or signal escalation criteria quickly expose gaps in alignment. Even when each local process appears compliant on paper, lack of consistency across regions signals weak global oversight rather than justified local variation.
As a result, inspectors frequently escalate findings when they detect fragmented safety decision-making. From a regulatory standpoint, inconsistency increases the risk of delayed signal detection, uneven patient protection, and unreliable post-marketing safety oversight.
How inspectors assess harmonization across global safety systems
Inspectors do not identify harmonization gaps through policy review alone; they recognize them when misalignment between regions translates directly into inspection findings.
They evaluate whether alignment exists not only in procedures, but also in data handling, decision logic, and escalation behavior under routine operating conditions.
This assessment focuses on the core areas inspectors use to judge alignment across global safety operations:
- Global safety governance and regulatory convergence
- Consistency of safety data and reporting practices
- Alignment of signal detection and management processes
- Controlled local flexibility within GMP and GVP frameworks
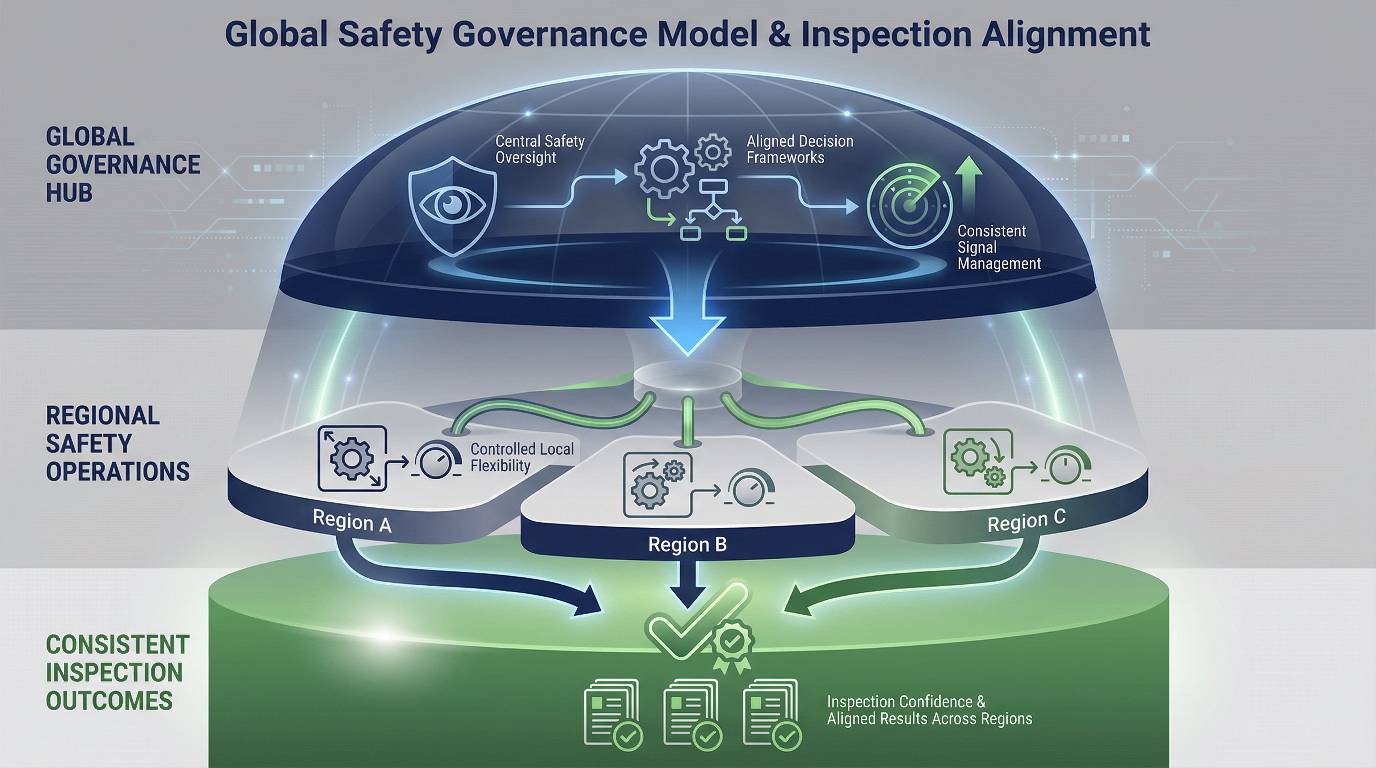
Global safety governance and regulatory convergence
Effective harmonization begins with governance. Inspectors review whether global safety leadership defines clear expectations that apply across affiliates, vendors, and partners. They assess whether oversight bodies actively monitor regional performance and intervene when deviations from global standards emerge.
In real inspections, governance weaknesses often surface when regional teams operate autonomously without meaningful global challenge. Inspectors interpret this as a lack of regulatory convergence, even when local compliance appears adequate.
Consistency of safety data and reporting practices
Data consistency is a central inspection focus. Regulators compare how safety data is collected, processed, and reported across regions. Differences in data completeness, coding practices, or reporting timelines indicate fragmented systems rather than controlled local adaptation.
Inspectors expect global safety systems to support uniform safety reporting consistency, ensuring that aggregated analyses reflect comparable inputs from all markets.
Alignment of signal detection and management processes
Signal detection alignment demonstrates whether harmonization extends beyond individual cases. Inspectors review how signals are identified, validated, and escalated globally. When regional teams apply different thresholds or decision criteria, regulators question the reliability of global signal management.
Aligned signal processes reassure inspectors that emerging risks are identified and addressed consistently, regardless of geographic origin.
Controlled local flexibility within GMP and GVP frameworks
Regulators accept local flexibility when it is controlled and justified. Inspectors therefore assess whether organizations clearly document where and why regional adaptations exist within GMP and GVP alignment expectations.
Uncontrolled variation, by contrast, is viewed as non-harmonized practice. Inspectors distinguish justified local requirements from discretionary deviations that weaken global safety oversight.
The role of international guidelines in driving alignment
Regulators evaluate harmonization through governance outcomes, not through the presence of global procedures alone.
Rather than enforcing uniform procedures, global standards provide a common reference point for safety governance, data integrity, and decision-making.
Inspectors assess how well organizations translate these expectations into operational alignment. They look for evidence that global standards inform local processes, training, and oversight without being applied mechanically. When companies rely solely on guideline citations without demonstrating operational convergence, inspectors often remain unconvinced.
Effective alignment emerges when international standards drive consistent safety behavior, not just consistent documentation.
Operational challenges companies face when harmonizing systems
Harmonization presents practical challenges that inspectors recognize but do not excuse.
Legacy systems often limit data integration, making it difficult to achieve consistent safety reporting. Regional autonomy can also complicate alignment when local teams resist centralized oversight.
Additionally, differing regulatory timelines and national requirements create operational friction. Inspectors understand these constraints, but they expect organizations to manage them proactively through governance, system integration, and clear accountability.
Where companies fail to address these challenges, inspection findings frequently cite fragmented safety systems, inconsistent decision logic, and weak global control.
Final Word
Recent multi-region inspection summaries suggest that a significant proportion of escalated pharmacovigilance findings—commonly cited in the 25–35% range—relate directly to global misalignment rather than isolated local failures, reinforcing that harmonization in pharmacovigilance is treated as a core compliance expectation. Regulators increasingly interpret inconsistent global safety behavior as evidence of insufficient control over post-marketing risk.
Organizations that invest in aligned governance, consistent safety execution, and controlled local flexibility reduce inspection risk and protect regulatory timelines. In practice, warning signs of weak harmonization often appear long before inspections, through repeated regional discrepancies that remain unresolved at the global level.
FAQs
Inspectors look for consistent safety decision-making, aligned reporting practices, and effective global oversight that produces comparable outcomes across regions under routine pharmacovigilance operations.
Because inconsistent safety practices signal weak global governance, causing regulators to question whether post-marketing risks are controlled uniformly across markets.
By aligning core safety principles, decision criteria, and escalation pathways while clearly documenting justified local adaptations within global governance frameworks.

Mahtab Shardi
Mahtab is a pharmaceutical professional with a Master’s degree in Physical Chemistry and over five years of experience in laboratory and QC roles. Mahtab contributes reliable, well-structured pharmaceutical content to Pharmuni, helping turn complex scientific topics into clear, practical insights for industry professionals and students.

Deviation Management in Pharma in 2026: GMP Process and Inspection Expectations
Deviation management plays a critical role in maintaining GMP compliance across pharmaceutical systems. This article explores how structured investigation, CAPA linkage, and quality risk management help organizations control deviations, reduce recurrence, and meet increasing inspection expectations.

Pharma Jobs in Kuwait in 2026: GCC Hiring and Compliance Insights
Kuwait’s pharmaceutical sector continues to expand under strict Ministry of Health regulations and GCC compliance frameworks. This guide explains regulatory pathways, major employers, salary expectations, and licensing requirements for professionals pursuing pharmaceutical careers within Kuwait’s growing Gulf healthcare market.

Critical Quality Attribute (CQA) in 2026: Expert Guide to Ensure Drug Quality
Critical Quality Attributes (CQAs) are crucial in pharmaceutical manufacturing because they directly affect drug safety, efficacy, and consistency. Regulatory agencies including the FDA and ICH require thorough CQA assessment to minimize risks and ensure high-quality medicines. By following best practices like early identification, risk assessment, and continuous monitoring, manufacturers can maintain robust production processes. Implementing modern strategies such as Quality by Design and integrating CQAs with quality assurance systems ensures compliance and safeguards patient health throughout the product lifecycle